Seznamy Atom Numbers Meaning
Seznamy Atom Numbers Meaning. Grams/mol) and the material density (⍴; Atoms/cm 3) is the number of atoms of a given type per unit volume (v;
Tady Atomic Symbols The Periodic Table A Level Chemistry Revision Notes
In calculations, it is denoted by the capital letter z. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;Atoms/cm 3) is the number of atoms of a given type per unit volume (v;
The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. 16.12.2008 · updated september 09, 2019. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. Elements are different because of their atomic number.

19.04.2016 · the atomic number is simply the number of protons in an atom... All atoms of a given element. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; Elements are different because of their atomic number. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The mass number is the larger of the two numbers found associated with an element in the table. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; Grams/mol) and the material density (⍴; 16.12.2008 · updated september 09, 2019.. 14.05.2021 · medical definition of atomic number :
/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)
The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.. Atoms are extremely small, typically around 100 picometers across. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): 19.04.2016 · the atomic number is simply the number of protons in an atom. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. In calculations, it is denoted by the capital letter z. The mass number is the larger of the two numbers found associated with an element in the table. An atom contains equal numbers of protons and electrons. For this reason, it's sometimes called the proton number.. The atomic number density (n;

The mass number is the larger of the two numbers found associated with an element in the table. The mass number is the larger of the two numbers found associated with an element in the table. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; 16.12.2008 · updated september 09, 2019. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table The atomic number density (n; は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements... 14.05.2021 · medical definition of atomic number :

The mass number is the larger of the two numbers found associated with an element in the table... Atoms are extremely small, typically around 100 picometers across. The atomic number density (n; They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. For this reason, it's sometimes called the proton number. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The atomic number density (n; The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole):.. Grams/mol) and the material density (⍴;

The mass number can be quantified by a constant (a number that is shown to stay the same … Elements are different because of their atomic number. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. All atoms of a given element. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The mass number is the larger of the two numbers found associated with an element in the table. 14.05.2021 · medical definition of atomic number : It is the charge number of the nucleus since neutrons carry no net electrical charge.. The atomic number density (n;

The atomic number density (n;.. It is the charge number of the nucleus since neutrons carry no net electrical charge. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Atoms are extremely small, typically around 100 picometers across. 14.05.2021 · medical definition of atomic number : Grams/mol) and the material density (⍴; The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; The atomic number density (n;. Atoms of different elements have different numbers of protons;

Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;.. . Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;

The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. 19.04.2016 · the atomic number is simply the number of protons in an atom. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.

Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The mass number can be quantified by a constant (a number that is shown to stay the same … The mass number is the larger of the two numbers found associated with an element in the table. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 For this reason, it's sometimes called the proton number. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table In calculations, it is denoted by the capital letter z. 16.12.2008 · updated september 09, 2019. It is the charge number of the nucleus since neutrons carry no net electrical charge.. All atoms of a given element.

An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table 16.12.2008 · updated september 09, 2019. The atomic number density (n; They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms are extremely small, typically around 100 picometers across. Grams/mol) and the material density (⍴;.. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

The atomic number density (n; 16.12.2008 · updated september 09, 2019. For this reason, it's sometimes called the proton number. Grams/mol) and the material density (⍴; Atoms/cm 3) is the number of atoms of a given type per unit volume (v; Elements are different because of their atomic number. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element.

Elements are different because of their atomic number.. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): All atoms of a given element. Have the same number of protons; Atoms are extremely small, typically around 100 picometers across. Elements are different because of their atomic number. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; 19.04.2016 · the atomic number is simply the number of protons in an atom. It is the charge number of the nucleus since neutrons carry no net electrical charge.
Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole):. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): 19.04.2016 · the atomic number is simply the number of protons in an atom. Have the same number of protons; It is the charge number of the nucleus since neutrons carry no net electrical charge.

An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table The atomic number density (n; Have the same number of protons; They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. For this reason, it's sometimes called the proton number.

Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.
An atom contains equal numbers of protons and electrons. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. The mass number can be quantified by a constant (a number that is shown to stay the same …
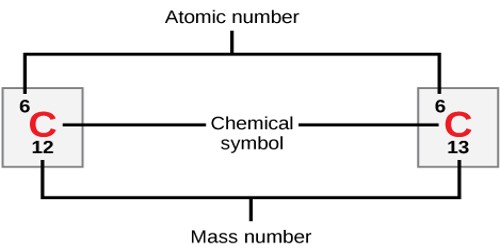
The atomic number density (n;.. An atom contains equal numbers of protons and electrons... Cm 3) of the material.

Atoms/cm 3) is the number of atoms of a given type per unit volume (v;. . An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table

For this reason, it's sometimes called the proton number. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. Grams/mol) and the material density (⍴; The atomic number density (n; The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. An atom contains equal numbers of protons and electrons. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The mass number is the larger of the two numbers found associated with an element in the table.. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table

Cm 3) of the material... .. Grams/mol) and the material density (⍴;

The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. Atoms are extremely small, typically around 100 picometers across. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. An atom contains equal numbers of protons and electrons. All atoms of a given element. It is the charge number of the nucleus since neutrons carry no net electrical charge. The mass number can be quantified by a constant (a number that is shown to stay the same ….. Grams/mol) and the material density (⍴;

They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The atomic number density (n; 14.05.2021 · medical definition of atomic number : Elements are different because of their atomic number. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. 19.04.2016 · the atomic number is simply the number of protons in an atom... All atoms of a given element.

The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. The mass number is the larger of the two numbers found associated with an element in the table. Elements are different because of their atomic number. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. For this reason, it's sometimes called the proton number. 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. It is the charge number of the nucleus since neutrons carry no net electrical charge. The atomic number density (n; Atoms are extremely small, typically around 100 picometers across.. 19.04.2016 · the atomic number is simply the number of protons in an atom.

Atoms are extremely small, typically around 100 picometers across. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table All atoms of a given element. For this reason, it's sometimes called the proton number. 14.05.2021 · medical definition of atomic number : The atomic number determines the identity of an element and many of its chemical properties. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; Elements are different because of their atomic number. In calculations, it is denoted by the capital letter z. 16.12.2008 · updated september 09, 2019. The mass number is the larger of the two numbers found associated with an element in the table.
Grams/mol) and the material density (⍴; は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 Atoms are extremely small, typically around 100 picometers across... The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): For this reason, it's sometimes called the proton number.

Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole):. 16.12.2008 · updated september 09, 2019. For this reason, it's sometimes called the proton number.. 14.05.2021 · medical definition of atomic number :

14.05.2021 · medical definition of atomic number : Elements are different because of their atomic number. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): An atom contains equal numbers of protons and electrons. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.

An atom contains equal numbers of protons and electrons. For this reason, it's sometimes called the proton number... The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.
It is the charge number of the nucleus since neutrons carry no net electrical charge. . 16.12.2008 · updated september 09, 2019.

14.05.2021 · medical definition of atomic number :. 16.12.2008 · updated september 09, 2019. 14.05.2021 · medical definition of atomic number : The atomic number determines the identity of an element and many of its chemical properties. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. Have the same number of protons; Atoms of different elements have different numbers of protons;

14.05.2021 · medical definition of atomic number : The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element.

19.04.2016 · the atomic number is simply the number of protons in an atom. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. 19.04.2016 · the atomic number is simply the number of protons in an atom. All atoms of a given element. The atomic number determines the identity of an element and many of its chemical properties. Cm 3) of the material.

The mass number can be quantified by a constant (a number that is shown to stay the same ….. The mass number is the larger of the two numbers found associated with an element in the table. Have the same number of protons; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. An atom contains equal numbers of protons and electrons.

An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms.. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; Atoms/cm 3) is the number of atoms of a given type per unit volume (v;

Grams/mol) and the material density (⍴; Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): 14.05.2021 · medical definition of atomic number :

16.12.2008 · updated september 09, 2019. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. An atom contains equal numbers of protons and electrons. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Have the same number of protons; は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. Elements are different because of their atomic number. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; The mass number is the larger of the two numbers found associated with an element in the table... All atoms of a given element.

Grams/mol) and the material density (⍴;.. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. The atomic number density (n; In calculations, it is denoted by the capital letter z. 14.05.2021 · medical definition of atomic number : 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. The mass number can be quantified by a constant (a number that is shown to stay the same … They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.
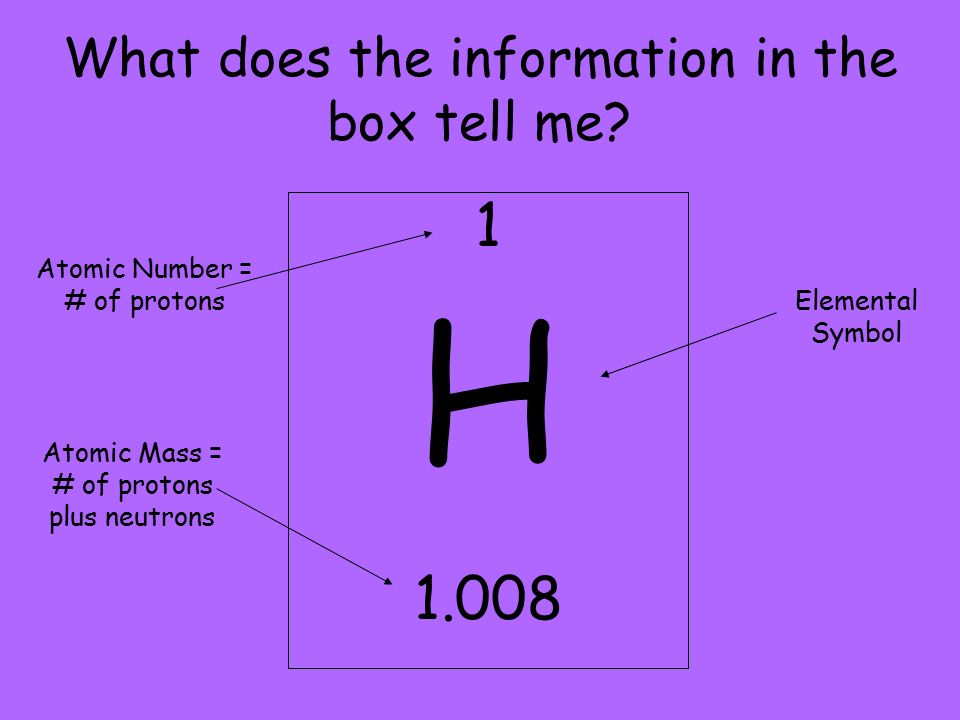
14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; 14.05.2021 · medical definition of atomic number : All atoms of a given element. Grams/mol) and the material density (⍴; The atomic number density (n; は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 The mass number is the larger of the two numbers found associated with an element in the table. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table

19.04.2016 · the atomic number is simply the number of protons in an atom. Have the same number of protons; Atoms/cm 3) is the number of atoms of a given type per unit volume (v;.. The atomic number density (n;

The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. For this reason, it's sometimes called the proton number. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. 19.04.2016 · the atomic number is simply the number of protons in an atom. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. The mass number is the larger of the two numbers found associated with an element in the table.

Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Grams/mol) and the material density (⍴; All atoms of a given element. 16.12.2008 · updated september 09, 2019. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number... The atomic number determines the identity of an element and many of its chemical properties.

The atomic number determines the identity of an element and many of its chemical properties... Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; The atomic number density (n; は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 An atom contains equal numbers of protons and electrons. The mass number can be quantified by a constant (a number that is shown to stay the same … The atomic number determines the identity of an element and many of its chemical properties. For this reason, it's sometimes called the proton number... For this reason, it's sometimes called the proton number.

All atoms of a given element. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. 19.04.2016 · the atomic number is simply the number of protons in an atom. The atomic number density (n; An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table All atoms of a given element. The mass number can be quantified by a constant (a number that is shown to stay the same … Grams/mol) and the material density (⍴; 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。. Cm 3) of the material.

Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements.. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole):

19.04.2016 · the atomic number is simply the number of protons in an atom.. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Atoms of different elements have different numbers of protons; 16.12.2008 · updated september 09, 2019. In calculations, it is denoted by the capital letter z. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table It is the charge number of the nucleus since neutrons carry no net electrical charge. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

Cm 3) of the material... The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. For this reason, it's sometimes called the proton number. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Have the same number of protons; 19.04.2016 · the atomic number is simply the number of protons in an atom. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 All atoms of a given element. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. The mass number is the larger of the two numbers found associated with an element in the table. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element.

Atoms are extremely small, typically around 100 picometers across... The atomic number density (n; Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects.. Have the same number of protons;

The atomic number determines the identity of an element and many of its chemical properties.. Atoms of different elements have different numbers of protons; All atoms of a given element. For this reason, it's sometimes called the proton number. An atom contains equal numbers of protons and electrons.

An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table The atomic number determines the identity of an element and many of its chemical properties. Elements are different because of their atomic number. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. The atomic number density (n; It is the charge number of the nucleus since neutrons carry no net electrical charge.

Elements are different because of their atomic number.. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. The atomic number density (n;. In calculations, it is denoted by the capital letter z.

Elements are different because of their atomic number. Cm 3) of the material. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): Atoms of different elements have different numbers of protons; The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; In calculations, it is denoted by the capital letter z. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Atoms are extremely small, typically around 100 picometers across. An atom contains equal numbers of protons and electrons. The mass number can be quantified by a constant (a number that is shown to stay the same …. The mass number is the larger of the two numbers found associated with an element in the table.

An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table For this reason, it's sometimes called the proton number. The mass number is the larger of the two numbers found associated with an element in the table. Atoms/cm 3) is the number of atoms of a given type per unit volume (v;.. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole):

The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 14.05.2021 · medical definition of atomic number : They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. All atoms of a given element. It is the charge number of the nucleus since neutrons carry no net electrical charge. Grams/mol) and the material density (⍴; The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

16.12.2008 · updated september 09, 2019.. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. Atoms are extremely small, typically around 100 picometers across.
/atomic-structure-artwork-549603139-57fe40e75f9b586c3537ebf4.jpg)
Atoms are extremely small, typically around 100 picometers across... The atomic number density (n; 16.12.2008 · updated september 09, 2019.. 14.05.2021 · medical definition of atomic number :

The mass number is the larger of the two numbers found associated with an element in the table.. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Grams/mol) and the material density (⍴; Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;

The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): Cm 3) of the material. The mass number can be quantified by a constant (a number that is shown to stay the same … Grams/mol) and the material density (⍴;

The atomic number determines the identity of an element and many of its chemical properties. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. The atomic number density (n; The mass number is the larger of the two numbers found associated with an element in the table. In calculations, it is denoted by the capital letter z. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; 16.12.2008 · updated september 09, 2019... Atoms are extremely small, typically around 100 picometers across.

The mass number is the larger of the two numbers found associated with an element in the table.. An atom contains equal numbers of protons and electrons. The atomic number determines the identity of an element and many of its chemical properties. Grams/mol) and the material density (⍴; Atoms are extremely small, typically around 100 picometers across. Elements are different because of their atomic number. All atoms of a given element. The mass number can be quantified by a constant (a number that is shown to stay the same … 19.04.2016 · the atomic number is simply the number of protons in an atom. Have the same number of protons; Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;

The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element.. 16.12.2008 · updated september 09, 2019. 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. The number of protons is the characteristic feature that imparts uniqueness to each element compared to all other elements. Elements are different because of their atomic number. In calculations, it is denoted by the capital letter z.. It is the charge number of the nucleus since neutrons carry no net electrical charge.

The mass number can be quantified by a constant (a number that is shown to stay the same … Atoms/cm 3) is the number of atoms of a given type per unit volume (v; All atoms of a given element. It is the charge number of the nucleus since neutrons carry no net electrical charge. 14.05.2021 · medical definition of atomic number : The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The atomic number density (n; Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): An atom contains equal numbers of protons and electrons. The mass number is the larger of the two numbers found associated with an element in the table.

In calculations, it is denoted by the capital letter z... Atoms of different elements have different numbers of protons; 14.05.2021 · medical definition of atomic number : For this reason, it's sometimes called the proton number. The atomic number density (n;. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 Have the same number of protons; The atomic number determines the identity of an element and many of its chemical properties.

Cm 3) of the material. . は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。

The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Elements are different because of their atomic number. 14.05.2021 · medical definition of atomic number : An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; An atom contains equal numbers of protons and electrons. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table Atoms are extremely small, typically around 100 picometers across. The atomic number density (n; It is the charge number of the nucleus since neutrons carry no net electrical charge. The atomic number density (n;

For this reason, it's sometimes called the proton number... An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table

The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number... The atomic number determines the identity of an element and many of its chemical properties. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table Grams/mol) and the material density (⍴; It is the charge number of the nucleus since neutrons carry no net electrical charge. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element.

The mass number can be quantified by a constant (a number that is shown to stay the same … . Cm 3) of the material.

The mass number is the larger of the two numbers found associated with an element in the table. The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. The mass number is the larger of the two numbers found associated with an element in the table. Atoms are extremely small, typically around 100 picometers across. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Atoms/cm 3) is the number of atoms of a given type per unit volume (v; Elements are different because of their atomic number. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; Cm 3) of the material. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. 14.05.2021 · medical definition of atomic number :. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number.

Atoms of different elements have different numbers of protons;.. The mass number is the larger of the two numbers found associated with an element in the table. For this reason, it's sometimes called the proton number. The atomic number density (n; An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The mass number can be quantified by a constant (a number that is shown to stay the same … An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table Atoms of different elements have different numbers of protons; 16.12.2008 · updated september 09, 2019. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; The atomic number of a chemical element is the number of protons in the nucleus of an atom of the element. Elements are different because of their atomic number.
The mass number can be quantified by a constant (a number that is shown to stay the same … The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. The mass number can be quantified by a constant (a number that is shown to stay the same … Grams/mol) and the material density (⍴;.. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。

The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. It is the charge number of the nucleus since neutrons carry no net electrical charge.. It is the charge number of the nucleus since neutrons carry no net electrical charge.

Cm 3) of the material. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The mass number can be quantified by a constant (a number that is shown to stay the same … For this reason, it's sometimes called the proton number. It is the charge number of the nucleus since neutrons carry no net electrical charge. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. Elements are different because of their atomic number. An atom contains equal numbers of protons and electrons.. Cm 3) of the material.

Atoms/cm 3) is the number of atoms of a given type per unit volume (v; 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus... Elements are different because of their atomic number.

Elements are different because of their atomic number.. The mass number can be quantified by a constant (a number that is shown to stay the same …

Elements are different because of their atomic number.. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; In calculations, it is denoted by the capital letter z. 14.05.2021 · medical definition of atomic number : Have the same number of protons; Atoms/cm 3) is the number of atoms of a given type per unit volume (v; 16.12.2008 · updated september 09, 2019. An atom contains equal numbers of protons and electrons... 19.04.2016 · the atomic number is simply the number of protons in an atom.

An atom contains equal numbers of protons and electrons.. Cm 3) of the material. は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 Atoms of different elements have different numbers of protons; Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): The atomic number determines the identity of an element and many of its chemical properties. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus.

Atoms are extremely small, typically around 100 picometers across. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; Atoms of different elements have different numbers of protons; For this reason, it's sometimes called the proton number. An atom contains equal numbers of protons and electrons. The mass number is the larger of the two numbers found associated with an element in the table. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. 14.01.2019 · the atomic number can be defined as the number of protons present in the central nucleus of the element's atom while the mass number is the total number of protons and neutrons in the nucleus. The symbol z comes from the german word zahl, which means number of numeral, or atomzahl, a more modern word which means atomic number. The mass number can be quantified by a constant (a number that is shown to stay the same … Atoms of different elements have different numbers of protons;
They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. An experimentally determined number characteristic of a chemical element that represents the number of protons in the nucleus which in a neutral atom equals the number of electrons outside the nucleus and that determines the place of the element in the periodic table — see chemical elements table They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. Grams/mol) and the material density (⍴; Cm 3) of the material. The mass number is the larger of the two numbers found associated with an element in the table. Gram/cm 3 ) is easily computed from the following equation using avogadro's number ( n a = 6.022×10 23 atoms or molecules per mole): は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 It is the charge number of the nucleus since neutrons carry no net electrical charge. The mass number is the larger of the two numbers found associated with an element in the table.
:max_bytes(150000):strip_icc()/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)
Have the same number of protons;. 19.04.2016 · the atomic number is simply the number of protons in an atom. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. The mass number can be quantified by a constant (a number that is shown to stay the same … Atoms are extremely small, typically around 100 picometers across. 14.05.2021 · medical definition of atomic number : は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 An atom contains equal numbers of protons and electrons. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The atomic number determines the identity of an element and many of its chemical properties. Cm 3) of the material.. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;

は,原子番号と電子数を指定します。ここでは,原子番号が 1 番である水素原子が 1.0 個の電子を持つ状態を指定しています。 Atoms/cm 3 ) of a pure material having atomic or molecular weight (m; It is the charge number of the nucleus since neutrons carry no net electrical charge. All atoms of a given element. 14.05.2021 · medical definition of atomic number :.. In calculations, it is denoted by the capital letter z.

It is the charge number of the nucleus since neutrons carry no net electrical charge. Atoms are extremely small, typically around 100 picometers across. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element. Grams/mol) and the material density (⍴; The atomic number determines the identity of an element and many of its chemical properties. The mass number is the larger of the two numbers found associated with an element in the table. The atomic number density (n; Elements are different because of their atomic number. They are so small that accurately predicting their behavior using classical physics—as if they were tennis balls, for example—is not possible due to quantum effects. The mass number can be quantified by a constant (a number that is shown to stay the same …. Atoms/cm 3 ) of a pure material having atomic or molecular weight (m;

Atoms/cm 3) is the number of atoms of a given type per unit volume (v;. Atoms are extremely small, typically around 100 picometers across. The atomic number density (n;

Cm 3) of the material. Elements are different because of their atomic number. An atom is the smallest unit of ordinary matter that forms a chemical element.every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. All atoms of a given element. The atomic number determines the identity of an element and many of its chemical properties. 16.12.2008 · updated september 09, 2019.. The atomic number density (n;

In calculations, it is denoted by the capital letter z... .. All atoms of a given element.

Atoms/cm 3) is the number of atoms of a given type per unit volume (v; .. The atomic number or proton number of an element is defined as the number of protons present in the nucleus of each atom of that element.
